
FDA’s transition to the Quality Management System Regulation (QMSR) isn’t just a paperwork exercise. While the alignment with ISO 13485 is old news, the real concern for manufacturers isn’t in the logic of risk – it’s in the evidence. Simply performing risk management activities is no longer enough: manufacturers must be able to show how risk‑based thinking actively shapes their quality system.
FDA will not be satisfied seeing risk buried in a static file – they want to see it operationalized. They’re asking: Is this control here because a spreadsheet said so three years ago, or because it’s actively protecting the patient today?
Although QMSR doesn’t change the fundamental requirement to manage risk, it completely changes how FDA evaluates your effectiveness. The old QSIT approach is being replaced by a more integrated, risk-based strategy outlined in Compliance Program 7382.850. Investigators are now trained to ask why certain processes are controlled the way they are, making your risk-based rationale the backbone of your defense.
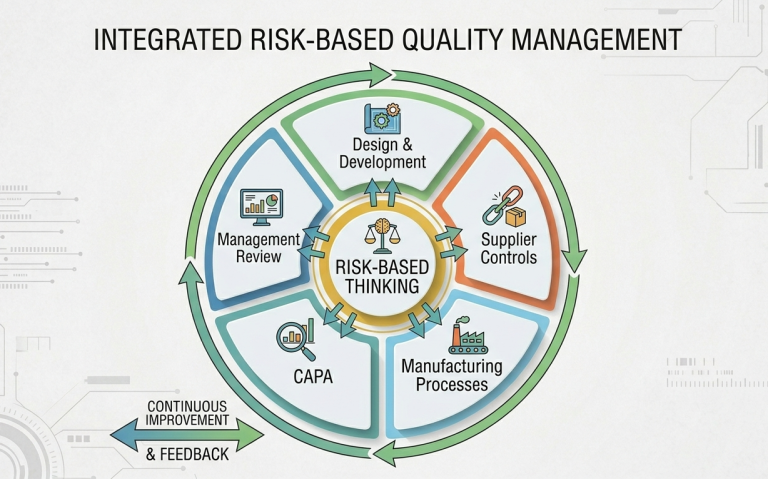
While ISO 13485:2016 Clause 4.1.2(b) has always required a risk-based approach to QMS processes, few organizations actually do it well. In a truly integrated system, risk considerations should be the primary driver behind decisions in manufacturing, supplier oversight, CAPA, and management review. QMSR (and CP 7382.850, by extension) is FDA’s way of pushing manufacturers out of their silos and toward a unified, system-wide strategy.
Most manufacturers have the basics: hazard analyses, risk files, and assessments. But QMSR asks a much tougher question: How do those assessments actually translate into day-to-day operations?
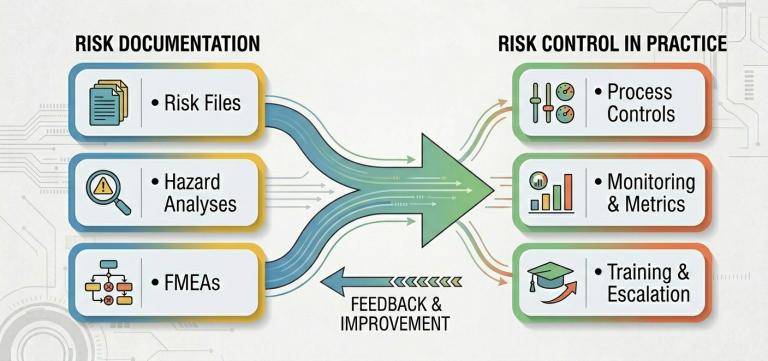
During an inspection, FDA will likely look for three things:
If your identified design risks don’t trigger specific controls in production or supplier oversight, your risk management isn’t a tool – it’s a static exercise. When those connections are weak, it becomes nearly impossible to defend your rationale to an investigator.
As organizations audit their QMSR readiness, we consistently see the same four friction points:
Under QMSR, “showing your work” means demonstrating how risk influences the path of the company. It’s about evidence, such as:
This matters, because FDA is looking at maturity. When an investigator asks why you prioritized one issue over another, a vague answer won’t cut it. You need a risk-based rationale that is baked into the structure of your operations.
Preparing for QMSR is more than a mapping exercise – it requires a fundamental gut-check:
Moving from “documentation” to “demonstration” is a heavy lift. ELIQUENT Life Sciences helps manufacturers make that move through:
Identify and address weaknesses before an inspection puts them under the spotlight. Reach out to ELIQUENT Life Sciences to learn how we can support your transition to a mature, risk-driven QMS.

US OfficeWashington DC
EU OfficeCork, Ireland


Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.

